Transforming Pathology Operations with efficiency, safety, and traceability
Turnaround
Traceability
Errors
(TAT)
With Authorization
Introduction
The Anatomic Pathology Module (AP) is a complete solution designed to empower histopathology, cytopathology, and surgical pathology operations within modern medical laboratories.
Developed to integrate seamlessly with LDM LIS and Superset BI, the AP Module elevates diagnostic accuracy, accelerates workflows, and enhances visibility across all phases — from specimen reception to final reporting.
Built with precision, automation, and compliance at its core, it transforms complex pathology processes into a smooth, traceable, and fully controlled digital workflow.


The Anatomic Pathology Module (AP)
is a complete solution designed to empower histopathology, cytopathology, and surgical pathology operations within modern medical laboratories.

Key Benefits
- Complete Workflow Automation
- Enhanced Diagnostic Accuracy
- Operational Efficiency
- Full Integration Across the Laboratory
- Compliance & Accreditation Readiness
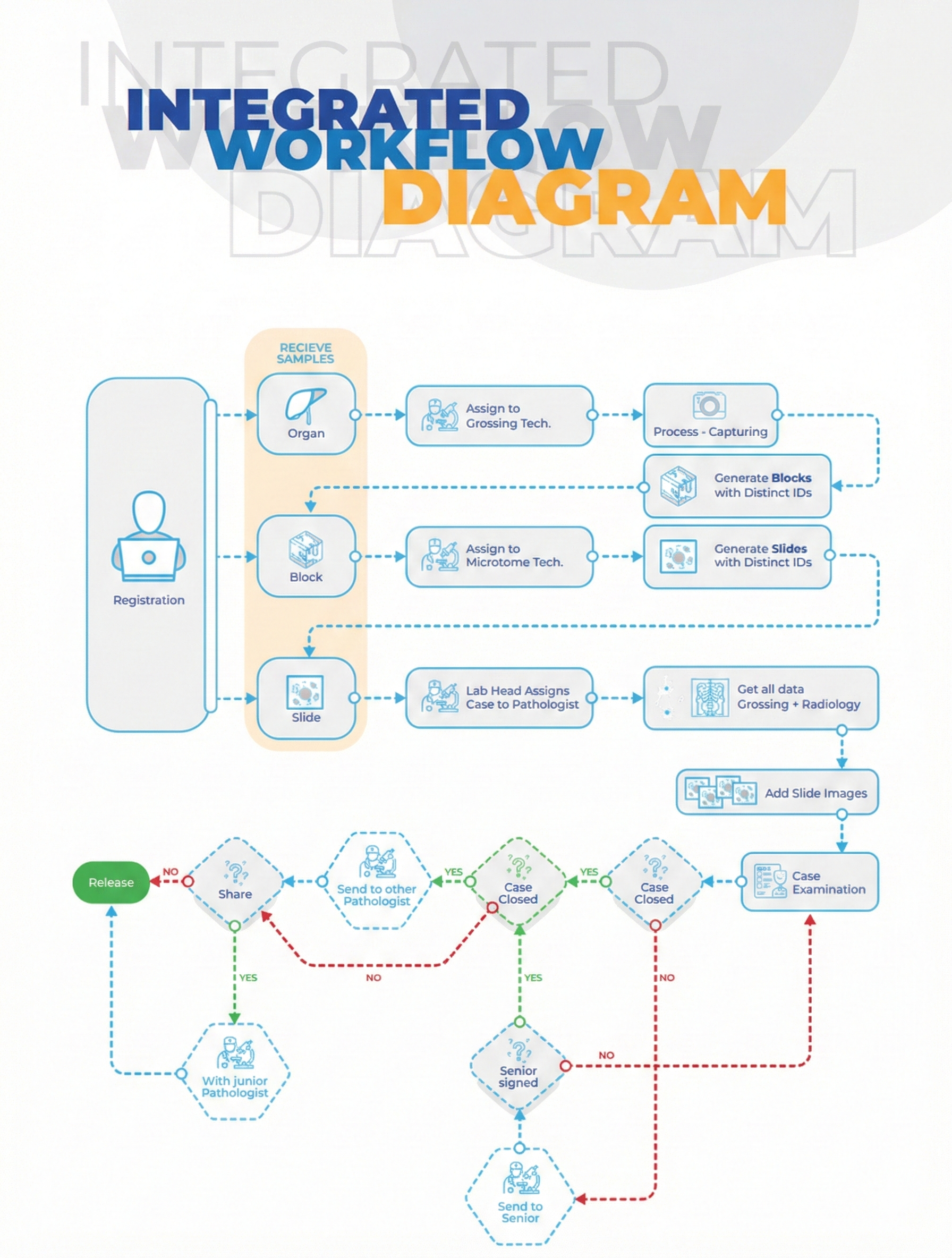
Workflow Diagram
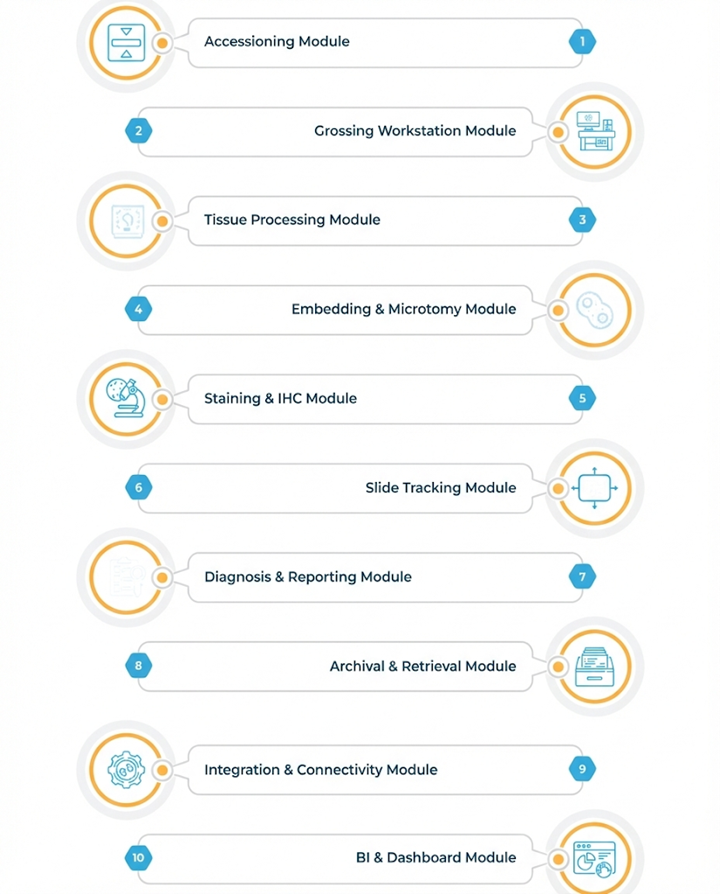
PMS Modules
Full list of Benefits
- Complete Workflow Automation: Eliminate manual processes from specimen reception to final reporting through a streamlined, digitally traceable pathway
- Enhanced Diagnostic Accuracy: Standardized templates, IHC scoring, and synoptic reporting ensure consistent and high-quality diagnostics
- Operational Efficiency: Barcode tracking and integrated device communication reduce errors and save valuable time
- Full Integration Across the Laboratory: Seamless connectivity with LDM Superset BI ensures a unified laboratory ecosystem
- Compliance & Accreditation Readiness :Designed to meet CAP, CLSI, and ISO 15189 standards with complete audit trails and traceability
- Intelligent Insights & BI Analytics: Integrated dashboards highlight TAT, productivity, utilization, IHC trends, and cancer registry data.
- Accurate Billing: Integration with LDM LIS ensures accurate billing and compliance with all regulations.
CORE FUNCTIONALITIES

- Specimen Management & Accessioning
• Unified accessioning for biopsies, resections, FNAC, PAP smears, and cytology samples.
• 2D barcoded labels for containers, cassettes, and slides.
• SNOMED CT & ICD-O coding integration.
• Digital photo capture and complete chain-of-custody tracking.
• Multi-specimen, multi-part case support.
- GROSSING WORKSTATION
• Dedicated grossing interface optimized for tablets and touch devices.
• Voice dictation and macro templates for reproducible descriptions.
• Cassette labeling and verification to eliminate mismatches.
• Specimen diagrams, measurements, margins, and weight recording.
- TISSUE PROCESSING
Batch assignment
It is the step where the system groups several pathology specimens into a batch based on shared criteria such as the instrument, test type, staining protocol, or processing cycle.
Real-time processing statusIt provides a moment-to-moment view of which samples are being processed, which samples are delayed, and what instruments are currently running.
Complete traceability of blocks throughout processingIt ensures every tissue block is accurately tracked throughout processing, preventing loss or mix-ups and protecting patient safety.
- EMBEDDING AND MICROTOMY
• Cassette-to-block tracking with technician identity stamping.
• Multi-slide labeling (routine, special stain, IHC).
• Levels, recuts, and unstained slide management.
• Slide distribution and pathologist assignment workflows.
- STAINING AND IHC MANAGEMENT
• Routine H&E, special stain, and immunohistochemistry (IHC) workflow management.
• Integration with automated stainers and Immunohistochemistry analyzers.
- SLIDE TRACKING SYSTEM
• Track every slide with full movement history.
• Status assignment (Pending, In Process, Under Review, Completed).
• Lost-slide prevention and location auditing.
• Complete visibility from microtomy to sign-out process.
- PATHOLOGIST WORKLIST AND DIAGNOSIS
• Smart dashboard for pending, urgent, and overdue cases.
• Template-driven microscopic descriptions.
• Integration with digital pathology systems.
• Support for multi-pathologist review and second opinions.
• Frozen section workflow with instant alerting.
• Integrated CAP/WHO synoptic reporting for key tumor types.
- FINAL REPORTING
• Rich-text editor for structured, narrative, and synoptic reports.
• Embedded images, diagrams, graphs, and IHC panels.
• Digital signatures and authorization hierarchy.
- STORAGE, ARCHIVAL AND RETRIEVAL
• Block and slide archival tracking with precise location mapping.
• Cabinet → Drawer → Box → Position coordinate system.
• Alerts for disposal deadlines.
Compliant with
Proven Quality ,global technology , global compliance





















