The Smartest Laboratory Solution for Operational Quality Management and Automated Daily Workflows
OUR NUMBERS TELL THE STORY
TURNAROUND TIME
AUDIT READINESS
(CES SCORE)
PRODUCTIVITY
OPERATIONAL COSTS
UTILIZATION SCORE
TRANSCRIPTION ERRORS
Introduction
The Smartest Laboratory Solution for Operational Quality Management and Automated Daily Workflows
Full automation and digital transformation of all stages and procedures of the daily work of the laboratory.
Keeps data secured, sorted and well-managed.
Managing a complicated daily workload in a simple, easy, accurate, and effective way.
Highly customizable and can be tailored to meet different types of laboratories, depending on the industry, regulations and workflows involved.


Serving 10 Industries
D'aliments et de Boissons Sector
Research Labs.
Oil and Gas
Water and De l'environnement
Criminal Evidence, Forensic
Fabrication Industry Sector
Pharmaceutical industry
La biotechnologie sector
L'Industrie Chimique
Genomic labs
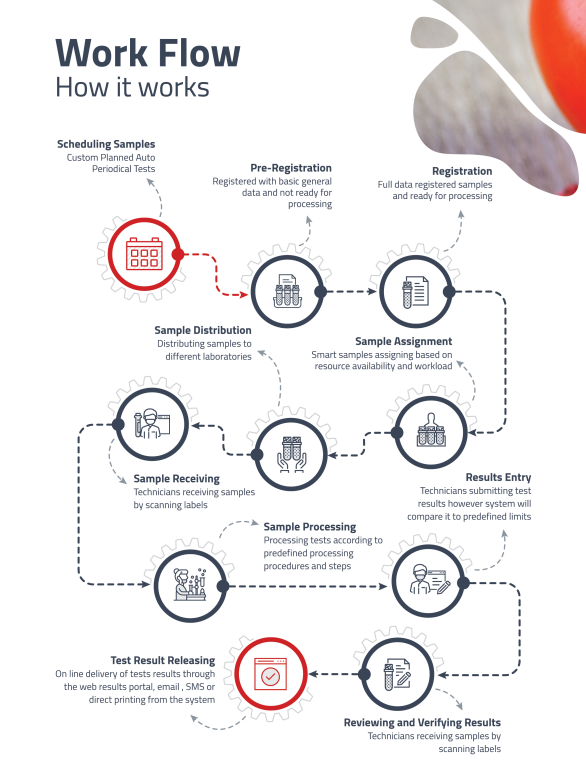
NT-LIMS Workflow
NT-LIMS Functions

CORE BUSINESS
-
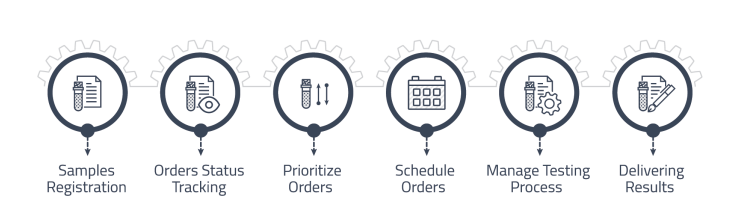
Order Management
Manage laboratory orders from clients and track them through the laboratory workflow.
 • Receiving orders from customers and registering samples.
• Receiving orders from customers and registering samples.
• Tracking status of each order from receipt to completion.
• Prioritizing orders based on customer requirements, sample type, and turnaround time.
• Ability to schedule orders based on laboratory workload and capacity.
• Pre-defined processing steps to guide and ensure tests are processed correctly.
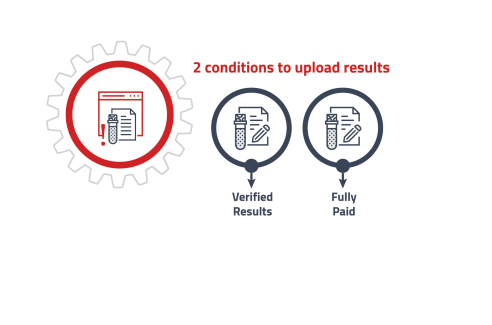
• Releasing ready results to customer with various delivery methods. -
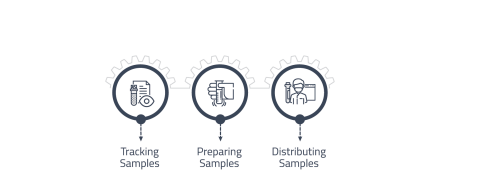
Sample Collection and Distribution
Accurately and efficiently collect and record information about samples that are submitted to the laboratory for testing.
 • Tracking the movement of samples and maintaining a complete chain of custody record.
• Tracking the movement of samples and maintaining a complete chain of custody record.
• Preparing samples for testing, including aliquoting and dilution.
• Distributing samples to the appropriate testing locations within the laboratory. -
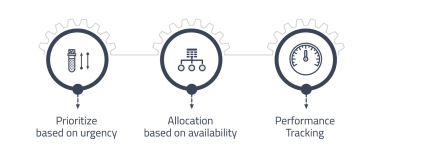
Workload Management
Efficiently manage and allocate laboratory resources such as personnel and equipment to maximize productivity.
 • Prioritizing tests based on urgency or criticality.
• Prioritizing tests based on urgency or criticality.
• Efficient resources allocation based on the availability of instruments and technicians.
• Tracking tests performance including turnaround time and accuracy. -
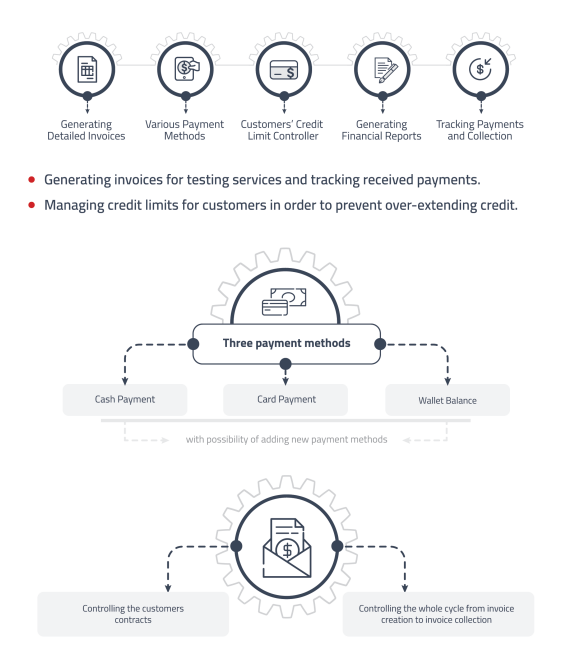
Customer Accounting
Streamline customer billing and invoicing processes with different payment methods and multiple price lists.
 • Generating invoices for testing services and tracking received payments.
• Generating invoices for testing services and tracking received payments.
• Managing credit limits for customers in order to prevent over-extending credit. -
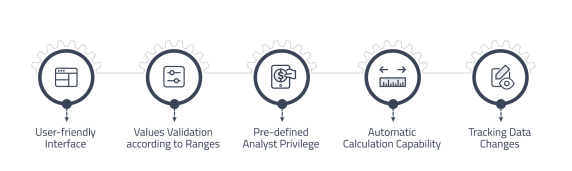
Results Entry and Review
Accurately record and manage test results in a centralized manner, ensuring data integrity and traceability.
 • Automated transfer of results from lab devices to the system.
• Automated transfer of results from lab devices to the system.
• User-friendly interface for manual result entry.
• Validating rules to ensure data falls within acceptable ranges.
• Results entry based on predefined authentication and privileges.
• Capability of automatic calculations and full change tracking. -
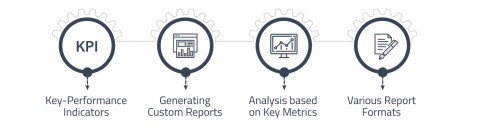
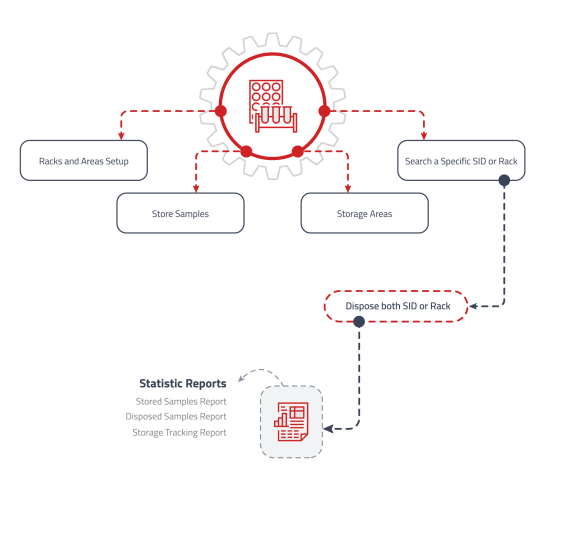
Statistics Reports
Allow laboratory managers to identify trends, track performance, and make data-driven decisions.
 • Display key performance indicators (KPIs) and relevant metrics.
• Display key performance indicators (KPIs) and relevant metrics.
• Analyze trends in test results and turnaround times.
• Export reports to various formats for stakeholders. -
Extra Modules: Precision, Accuracy, Efficiency, and Reliability
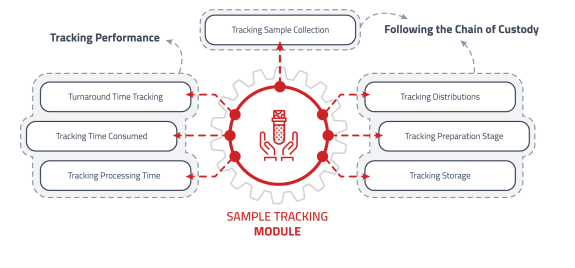
Sample Assignment and Tracking
Designed to manage and track the movement of samples within the laboratory.
 • Create sample records with unique identifiers.
• Create sample records with unique identifiers.
• Generate metrics related to processing and turnaround time.
• Manage chain of custody records for every sample handler. -
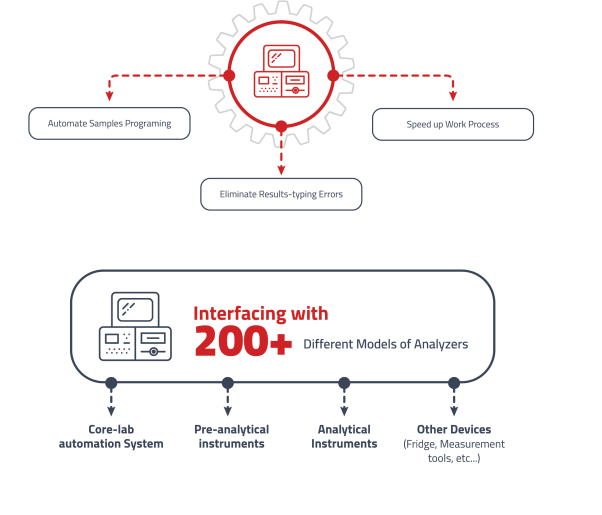
Equipment Interfacing Management
Direct communication between laboratory instruments and LIMS to eliminate manual entry.
 • Eliminating manual data entry to ensure data accuracy.
• Eliminating manual data entry to ensure data accuracy.
• Bidirectional communication with instruments in real-time.
• Support for a variety of instrument communication protocols.
• Automated validation against predefined rules. -
Sample Storage Management
Manage storage locations, optimize capacity, and ensure regulatory compliance.
 • Track exact locations including unit, shelf, and position.
• Track exact locations including unit, shelf, and position.
• Manage retrieval history and quantity tracking.
• Real-time location updates and notifications. -
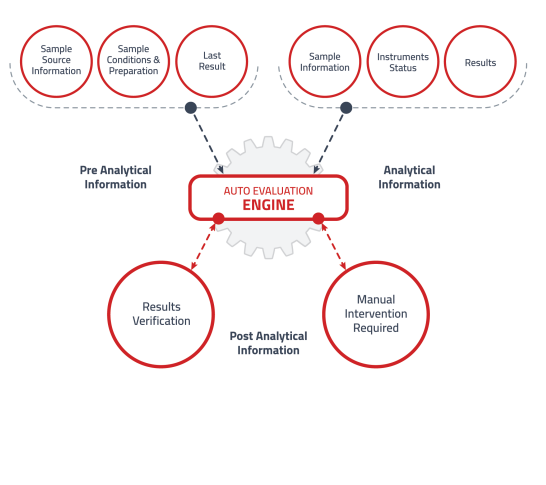
Auto-Evaluation Module
Reduce workload and improve accuracy through predefined evaluation rules and algorithms.
 • Significantly reduce time for result evaluation and release.
• Significantly reduce time for result evaluation and release.
• Minimize human error risk in the testing process.
• Fully customizable rules to suit specific laboratory needs. -
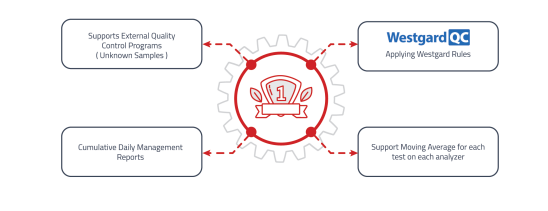
Quality Control Module
Critical function to ensure laboratory results meet internal and external quality standards.
 • Scheduling and tracking quality control tests.
• Scheduling and tracking quality control tests.
• Monitoring instrument performance with automated alerts.
• Long-term trend analysis for QC data patterns. -
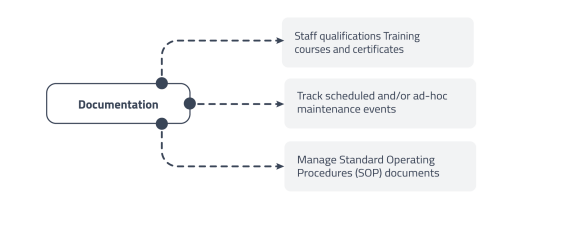
Quality Assurance and Documentation
Ensuring compliance with standards like ISO 17025 through rigorous documentation.
 • Store SOPs, test methods, and staff training records.
• Store SOPs, test methods, and staff training records.
• Manage calibration and maintenance schedules for all instruments.
• Framework for monitoring laboratory reliability and competency. -
Outsource Labs Manager
Manage relationships with third-party contract laboratories via a centralized platform.
• Manage sample flow and shipment tracking with external labs.
• Seamless data exchange for external test results.
• Cost and performance analysis for outsourced testing. -
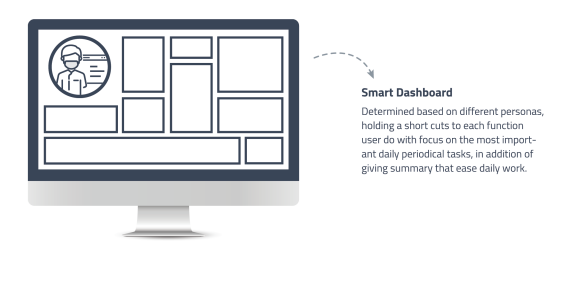
Dashboard Management
High-level graphical overview of laboratory operations to support informed decision-making.
 • Customizable dashboards for analysts, managers, and admins.
• Customizable dashboards for analysts, managers, and admins.
• Advanced visualizations including histograms and heat maps.
• Data mining to identify patterns and gain competitive advantages. -
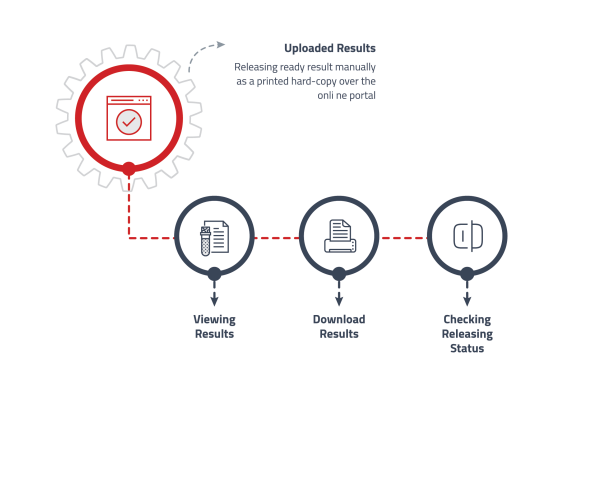
Results Portal
Timely and secure sharing of test results with customers while maintaining privacy.

 • Authenticated access based on login credentials.
• Authenticated access based on login credentials.
• Real-time access to test results as they become available.
• Interactive tools to analyze portal traffic and customer usage. -
Notifications Management
Real-time updates on sample status via SMS or E-mail for personnel and customers.
• Deliver critical messages regarding sample arrival or test completion.
• Automated alerts for results falling outside acceptable ranges.
• Emergency notifications for power outages or malfunctions.
• Improved overall functionality through instant communication.
Our Selected Clients
More than 2000+ Labs in 12 countries